How does CO2 warm the air and why it's a problem
Show how CO2 holds more energy than regular air.
Yield: 1 demo
Cost: 270
Equipment
- Tape
- Small piece of glass
Materials
- 2 bags zip lock
- 1 molecule kit https://www.amazon.com/dp/B01NCU854K/
- 1 infrared camera https://www.amazon.com/dp/B075F61GFH/
Instructions
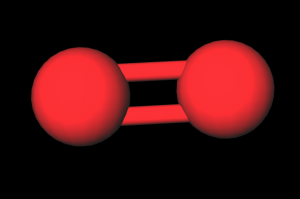
- Build O2 & N2: Assemble two atoms (red) with a rigid connection & assemble two blue atoms with a rigid connection
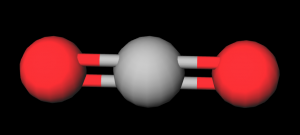
- Build CO2: Take a black atom and using the flexible
- Fill a zip lock bag by exhaling in to the bag. Zip up back to lock in some CO2 air, label it as CO2. Fill another bag with air and zip it up label it as Air.
Notes
Explain to the students that: As sunlight enters the earths atmosphere the light energy from the sun hits the surface of the earth and converts to heat energy with a low frequency. This energy is the captured by lighter bound molecules of CO2 and methane. These molecules then radiate heat back toward earth thus increasing the temperature of the earth.
Show the students how the oxygen and nitrogen molecules are rigid thus they don't absorb as much energy from the environment. Next show how the carbon dioxide molecule is flexible thus it can absorb the energy from the environment and the radiate it in all directions including back toward earth. Explain how heat causes the molecule to vibrate and that is how the energy is captured.
Next point the infrared camera at a student and place a the small piece of glass between the camera and the student. Ask the students what they see. The glass is blocking most of the infrared heat from being captured by the camera. Now place the bag labelled air between the student and the camera. Ask them what they see and why they are seeing that. Next place the bag of CO2 between the student and the camera. Ask them what they see and why. They should see that the bag labelled CO2 let less energy through because it was absorbing the heat energy from the student. The bag labeled air absorbed less.
Additional Questions:
- What are some sources of carbon dioxide and methane?
- What are ways to limit carbon dioxide?
- How can we remove carbon dioxide?